Neuren Pharmaceuticals Limited (ASX: NEU) has advised that its licensing partner, Acadia Pharmaceuticals (Nasdaq: ACAD), will request a re-examination of a negative opinion issued by the Committee for Medicinal Products for Human Use (CHMP) regarding trofinetide in Europe.

Neuren Pharmaceuticals Limited confirmed its partner Acadia Pharmaceuticals will seek a CHMP re-examination following a European regulatory setback. [Bioshares]
The pharmaceutical regulatory update follows the CHMP’s formal vote to refuse the Marketing Authorisation Application (MAA) for trofinetide for the treatment of Rett syndrome in patients aged two years and older. The application was submitted to the European Medicines Agency (EMA).
The development is significant for Neuren investors given trofinetide’s established commercial footprint in approved markets and Europe’s substantial unmet need in Rett syndrome.
Key Findings and Regulatory Context
Trofinetide, marketed as DAYBUE® in approved regions, remains authorised in the United States, Canada and Israel. It is currently the first and only approved therapy for Rett syndrome in those jurisdictions.
Trofinetide remains approved in multiple markets as the first authorised treatment for Rett syndrome. [Business Wire]
According to Acadia, the CHMP’s refusal was based on several perceived limitations in the pivotal LAVENDER clinical trial data. These included:
- The magnitude of the treatment effect at 12 weeks is being considered limited
- The view that not all core symptoms of Rett syndrome were captured in trial endpoints
- Concerns regarding longer-term outcome assessments influenced by patient discontinuations
Despite these concerns, the LAVENDER trial met its co-primary and key secondary endpoints. Management maintains that the data demonstrate clinically meaningful benefits for patients.
In approved markets, trofinetide has been available for nearly three years, supporting its commercial viability. Compared with global benchmarks in rare neurological disorders, having regulatory clearance in multiple advanced markets strengthens its evidence base and revenue stability.
Economic and Strategic Implications
The pharmaceutical regulatory update arrives at a time when rare disease treatments continue to command premium pricing and attract investor attention. Global demand for therapies targeting neurodevelopmental and genetic disorders remains strong, supported by orphan drug incentives and streamlined regulatory pathways.
Acadia’s Chief Executive Officer, Catherine Owen Adams, stated that while the Company is disappointed with the CHMP recommendation, it remains encouraged by the therapy’s demonstrated benefits and strong engagement from patients and clinicians. She confirmed the Company’s commitment to working constructively with EU regulators through the re-examination process.

Acadia’s Chief Executive Officer, Catherine Owen Adams. [ACADIA]
Neuren Chief Executive Officer Jon Pilcher added that trofinetide has been making a difference for patients in approved markets and that the unmet medical need in Europe remains substantial and urgent.
For investors, the re-examination represents both regulatory risk and potential upside. A successful outcome would open access to the European market, expanding revenue opportunities for Acadia and generating milestone and royalty flows for Neuren under its exclusive worldwide licence agreement.
Resource Pipeline and Clinical Development Update
Neuren retains a diversified pipeline beyond trofinetide. The Company’s second drug candidate, NNZ-2591, is in clinical development for multiple neurodevelopmental disorders.
Key programs include:
- Phase 2 trials completed in Phelan-McDermid syndrome, Pitt-Hopkins syndrome and Angelman syndrome
- Orphan drug designation in the United States and the European Union
- Fast Track and Rare Pediatric Disease designations from the U.S. Food and Drug Administration
Neuren is also conducting a Phase 3 randomised, double-blind, placebo-controlled trial known as “Koala” evaluating NNZ-2591 in children aged three to twelve years with Phelan-McDermid syndrome. A 52-week open-label extension study is underway.
The Company’s development approach targets the role of insulin-like growth factor 1 (IGF-1) in the brain using orally administered analogues of naturally occurring peptides. Its focus on rare and genetically defined neurological conditions positions it within high-need, lower-competition therapeutic categories.
Market and Industry Context
The global rare disease pharmaceutical sector continues to expand, supported by regulatory incentives and growing diagnostic capabilities. Europe remains a key commercial region, though regulatory scrutiny from EMA committees such as the CHMP can introduce timing uncertainty.
For Acadia Pharmaceuticals, which is listed on the Nasdaq exchange, the re-examination process represents a strategic effort to address regulatory concerns and potentially secure approval without initiating a new full-scale clinical program. For Neuren, whose shares last traded at $12.560 with a market capitalisation of approximately $1.74 billion, developments around trofinetide remain central to valuation metrics.
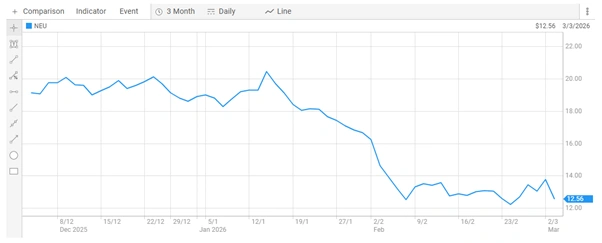
Neuren Pharmaceuticals Limited Share Price [ASX]
The re-examination procedure allows the sponsor to respond formally to the CHMP’s objections, submit clarifications, and seek reconsideration. While outcomes are not guaranteed, the pathway provides a defined regulatory mechanism to contest the opinion.
Investors and analysts will now monitor timelines for the re-examination request, potential additional data submissions, and feedback from European regulators. The outcome will shape Neuren’s medium-term revenue outlook and influence broader sentiment toward rare neurological drug approvals in the EU.
As the pharmaceutical regulatory update unfolds, market participants are likely to balance near-term regulatory risk against the established commercial performance of trofinetide in approved markets and the strength of Neuren’s broader clinical pipeline.