Dimerix Limited (ASX: DXB) has invited investors to attend two in-person briefings in Perth and Singapore during March and April 2026. The company will provide a clinical and commercial update on its lead drug candidate, DMX-200, and outline its broader growth strategy.
The Melbourne-based biopharmaceutical group confirmed the events on 4 March 2026. Chief Executive Officer and Managing Director Dr Nina Webster will lead the presentations. The sessions will focus on the global Phase 3 ACTION3 study in focal segmental glomerulosclerosis, a rare kidney disease with no specifically approved therapies in the United States.

Dr Nina Webster to lead Dimerix 2026 investor briefings in Perth and Singapore. [Euroz Hartleys]
Perth Investor Briefing Targets Existing and Prospective Shareholders
Dimerix will host the Perth investor briefing on Monday, 16 March 2026. The session will begin at 1:15 pm AWST, with arrivals from 1:00 pm. The venue is Steves Bar & Café, 30 The Avenue, Nedlands.

Perth briefing location for Dimerix investor session, 16 March 2026. [Star Wine list]
Dr. Webster will present an update on the ACTION3 Phase 3 clinical trial and discuss the commercial partnering status of DMX-200. Investors will also have the opportunity to ask questions during the session. The company stated that light refreshments will be served.
Attendance is limited. Investors must RSVP to the IR Department by Thursday, 12 March 2026. The dress code is smart casual or business attire.
Singapore Joint Briefing Highlights Regional Engagement
Dimerix will hold a second briefing in Singapore on Monday, 20 April 2026. The event will begin at 12:30 pm SGT, with arrivals from 12:15 pm. The company confirmed that the venue will be located in the Singapore CBD, with final details to follow.
Dr Nina Webster and Chief Operating Officer Dr Rob Shepherd will represent Dimerix. Dr Daniel Tillett, CEO and Managing Director of Racura Oncology, will also present. Both companies will outline their development strategies and progress.
The Singapore session will include lunch. Attendees must advise of dietary requirements when submitting their RSVP. Registrations close on Friday, 20 March 2026.
ACTION3 Phase 3 Trial Update in FSGS Kidney Disease
Dimerix will centre both briefings on its global ACTION3 Phase 3 study. The trial evaluates DMX-200 in patients with focal segmental glomerulonephritis (FSGS).
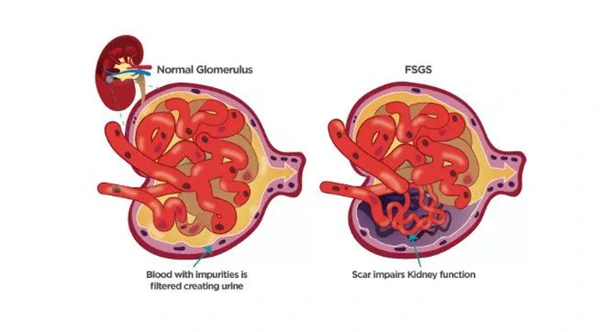
DMX-200 targets inflammatory pathways in FSGS kidney disease. [Medizzy]
The ACTION3 study is a multi-centre, randomised, double-blind, placebo-controlled trial. It tests the efficacy and safety of DMX-200 in patients who already receive a stable dose of an angiotensin II receptor blocker. These medicines form part of the current standard of care for hypertension and kidney disease.
After stabilisation on background therapy, participants receive either DMX-200 at 120 mg twice daily or a placebo. The treatment period runs for two years. The trial aims to measure reductions in proteinuria and changes in kidney function using the estimated glomerular filtration rate slope.
Dimerix designed the study to generate evidence that could support marketing approval. Further study details appear on ClinicalTrials.gov under identifier NCT05183646 and on the Australian New Zealand Clinical Trials Registry under ACTRN12622000066785.
DMX-200 Commercial Strategy and Orphan Drug Status
DMX-200 acts as a chemokine receptor CCR2 antagonist. Patients take the therapy alongside an angiotensin II type I receptor blocker. The company stated that this combination approach targets inflammatory pathways linked to kidney injury.
Dimerix has secured granted patents for DMX-200 in various territories until 2032. Patent applications submitted globally may extend protection until 2045. The drug has also received Orphan Drug Designation in the United States, Europe, the United Kingdom and Japan.
Orphan Drug Designation can provide regulatory and commercial incentives, including market exclusivity after approval in certain jurisdictions. The company indicated that it continues to pursue commercial partnering discussions as part of its global strategy.
FSGS Market Need and Clinical Background
FSGS is a rare and serious kidney disorder characterised by progressive scarring in parts of the glomeruli. The condition leads to proteinuria and declining kidney function. Many patients progress to end-stage renal disease.
In the United States, more than 40,000 people live with FSGS, including adults and children. Currently, no therapies have received specific approval for FSGS in the U.S. Physicians rely on non-specific immunosuppressive and supportive treatments.
Patients with progressive or treatment-resistant disease can reach end-stage kidney disease within five years of diagnosis. Even after kidney transplantation, recurrence occurs in up to 60 per cent of cases. These figures highlight the need for new treatment options.
Researchers now recognise that inflammation contributes to disease progression. Monocyte and macrophage activation play a role in glomerular injury. Dimerix designed DMX-200 to target relevant inflammatory pathways.
Also Read: Neuren Pharma European Regulatory Update on Trofinetide
Company Outlook and Forward Statements
Dimerix authorised the release of the briefing invitation through Dr Nina Webster. The company stated that it believes the briefings will provide investors with direct access to management and current clinical information.
The release included forward-looking statements regarding clinical development, regulatory processes and commercial plans. Management cautioned that actual outcomes may differ due to risks associated with drug development, manufacturing, regulatory approvals and capital requirements.
Dimerix trades on the Australian Securities Exchange under the ticker DXB. The company continues to focus on advancing DMX-200 through late-stage clinical development while exploring partnership opportunities.
Investors seeking further information can contact the company directly or visit its official website. The Perth and Singapore briefings will offer stakeholders an opportunity to engage with executives and review the latest progress in the Phase 3 FSGS program.