CLINUVEL Pharmaceuticals (ASX: CUV) launched its expansive program at the American Academy of Dermatology (AAD) Annual Meeting in Denver on 27–31 March 2026, showcasing a proprietary AI-driven vitiligo assessment tool alongside its SCENESSE® (afamelanotide) program for erythropoietic protoporphyria (EPP).
The Melbourne-based Company introduced the Vitiligo Visual Algorithm (VVA) to an audience of over 20,000 dermatology professionals, marking a major step in its global clinical strategy.

CLINUVEL Pharmaceuticals showcases its Pavilion of Photomedicine at the 2026 American Academy of Dermatology Annual Meeting in Denver. [LinkedIn]
CLINUVEL Unveils AI-Driven Vitiligo Assessment at AAD
CLINUVEL presented a broad program across scientific sessions, exhibitions, and industry events at the 2026 AAD Annual Meeting, the largest dermatology congress in North America.
A key highlight was the launch of the Vitiligo Visual Algorithm (VVA), an in-house AI tool designed to assess pigmentation changes using standardised clinical photography objectively. The system aims to improve how physicians track treatment progress and make clinical decisions.
Dr. Linda Teng stated the Company is working toward establishing a new standard in vitiligo assessment through data-driven insights.
Fact Box:
- 20,000+ dermatology professionals attending AAD 2026
- Source: CLINUVEL ASX Announcement (27 March 2026)
The Vitiligo Visual Algorithm (VVA) uses AI to assess pigmentation changes from clinical photographs.[Skin Horizon]
Why This Development Matters
The introduction of the VVA addresses a long-standing challenge in dermatology: the lack of objective, standardised tools for measuring vitiligo progression. By applying AI to clinical imaging, CLINUVEL aims to reduce subjectivity in both trials and real-world treatment.
Strategically, the Company is strengthening its dual focus: advancing vitiligo therapies while expanding its commercial EPP treatment. The VVA could also become a proprietary advantage if integrated into clinical trials and widely adopted.
However, regulatory validation and clinical outcomes remain key uncertainties.
Companies and Organisations Involved
CLINUVEL Pharmaceuticals Limited is a Melbourne-headquartered specialty pharmaceutical group focused on photomedicine and melanocortin therapies. Its lead product, SCENESSE® (afamelanotide 16mg), is approved in multiple regions as the first systemic photoprotective drug.
Other stakeholders include the Global Vitiligo Foundation and additional dermatology societies contributing to scientific discussions and feedback during the event.
Global Stage: AAD 2026 in Denver
The AAD Annual Meeting is being held in Denver from 27 to 31 March 2026. CLINUVEL’s presence includes a 4,000-square-foot Pavilion of Photomedicine, designed as an immersive exhibition space.
Although hosted in the United States, the event draws international participants, reflecting the global relevance of CLINUVEL’s therapies and pipeline.
Timeline of Key Developments
- 2025: Development of the VVA begins
- 26 March 2026: Program data presented at satellite symposia
- 27 March 2026: Official unveiling at AAD and ASX announcement
- 27–31 March 2026: Ongoing presentations and live demonstrations
This builds on CLINUVEL’s growing presence at AAD, following a successful 2025 showing.
Market Snapshot: CLINUVEL Pharmaceuticals (ASX: CUV)
Last traded price: $9.860
Daily change: +$0.080 (+0.817%)
Trading volume: 39,294 shares
Bid–offer range: $9.860 – $9.880
Market capitalisation: ~$490.96 million
Market segment: Mid-cap specialty pharmaceutical company
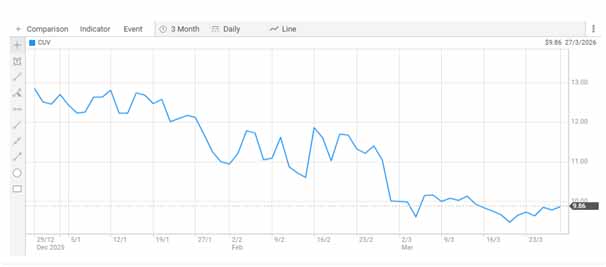
CLINUVEL Pharmaceuticals Share price [ASX]
Outlook: What Comes Next
CLINUVEL plans to integrate the VVA into its upcoming Phase III vitiligo trial (CUV107), which could support future regulatory submissions if successful. At the same time, the Company continues expanding awareness and adoption of SCENESSE® for EPP.
Key risks include regulatory acceptance of AI-based clinical tools and the broader challenges associated with drug development.
Also Read: Catapult Sports FY26 ACV Growth Forecast Hits 28% – Colitco
Final Thoughts
CLINUVEL’s AAD 2026 program reflects a clear strategy: combine commercial strength in rare diseases with innovation in AI-driven dermatology. If validated, the Vitiligo Visual Algorithm could play a pivotal role in shaping how vitiligo is assessed and treated globally.
FAQs
Q1. What did CLINUVEL announce at AAD 2026?
CLINUVEL Pharmaceuticals Limited unveiled its Vitiligo Visual Algorithm (VVA), an AI-driven tool for assessing pigmentation changes, alongside updates on its SCENESSE® therapy at the American Academy of Dermatology Annual Meeting in Denver.
Q2. What is the Vitiligo Visual Algorithm (VVA)?
The VVA is a proprietary AI-based system that analyses standardised clinical photographs to objectively measure skin pigmentation changes in vitiligo patients, improving treatment tracking.
Q3. Why is the VVA important for dermatology?
It introduces a standardised, data-driven method for evaluating vitiligo, addressing the long-standing challenge of subjective assessment in both clinical trials and practice.
Q4. What is SCENESSE® used for?
SCENESSE® (afamelanotide) is used to treat erythropoietic protoporphyria (EPP), a rare condition that causes extreme sensitivity to sunlight, and is currently the only approved systemic therapy for it.
Q5. What are the next steps for CLINUVEL’s vitiligo program?
The Company plans to integrate the VVA into its Phase III CUV107 trial, which could support future regulatory approval if clinical outcomes are positive.
Q6. Where is the AAD 2026 meeting being held?
The 2026 AAD Annual Meeting is being held in Denver from 27 to 31 March 2026, with over 20,000 dermatology professionals expected to attend.
Disclaimer:
This article is published by Colitco for informational purposes only and does not constitute financial, investment, or medical advice. The information is based on publicly available disclosures from CLINUVEL Pharmaceuticals Limited and other sources believed to be reliable at the time of writing. Colitco does not guarantee the accuracy, completeness, or timeliness of the information and accepts no liability for any decisions made based on this content. Readers should conduct their own independent research or consult a qualified professional before making any investment or healthcare decisions.
Sources
https://www.asx.com.au/markets/company/CUV