Neuren Pharmaceuticals Limited (ASX: NEU) released its Neuren investor presentation 2026 on 27 February 2026. The update covered three things: a growing commercial royalty stream from DAYBUE, a maturing NNZ-2591 clinical pipeline and a strong cash position to fund both.

Figure 1: Neuren Pharmaceuticals Limited headquarters and branding signage [LinkedIn]
The Company is no longer a pure-play development-stage biotech. Neuren Pharmaceuticals ASX is generating real profit from an approved drug while simultaneously running multiple late-stage clinical programs across serious childhood neurological conditions that currently have no approved treatments.
Neuren Financial Highlights Show Rising Profitability and Cash Strength
The Neuren financial highlights from the FY2025 result underline the Company’s transition to a commercially funded model:
- Royalty income up 15% year-on-year to $65 million
- Interest income of $12 million
- NPAT of $30 million, excluding one-time revenue items
- Corporate and administration costs of only $6 million
- R&D investment in NNZ-2591 of $36 million
- Cumulative DAYBUE income since launch in 2023: $510 million
- Cash position of $296 million as at 31 December 2025, up from $222 million at 31 December 2024
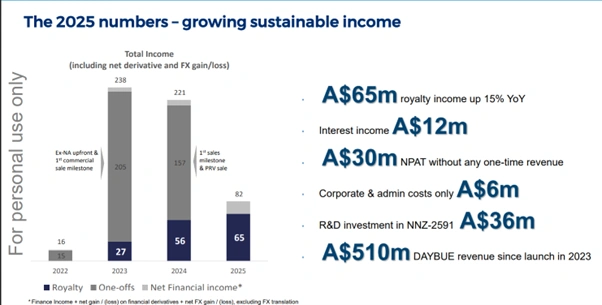
Figure 2: Neuren’s FY2025 financial performance showing rising royalty income, profitability and R&D investment [Neuren Pharmaceuticals]
The cash bridge tells a clean story. Royalty income contributed $63 million, and the one-time sales milestone and Priority Review Voucher sale added $159 million. After tax, interest, operating payments and capital management, the Company closed the year with $296 million in cash.
DAYBUE Performance in the Neuren Investor Presentation 2026
DAYBUE (trofinetide), commercialised by Acadia Pharmaceuticals in the US, delivered US$391 million in net sales in CY2025, up 12% on CY2024. In the Neuren financial highlights, Acadia has guided CY2026 net sales of US$460 million to US$490 million, representing growth of 18-25%.
Key DAYBUE operating metrics from the presentation:
- 1,070 patients on active treatment as of Q4 2025, up from 920 in Q4 2024
- Treatment persistence at 12 months is approximately 55%
- Discontinuations remain in the low single digits per quarter
- Approximately 76% of new prescriptions are now coming from community settings rather than specialist centres
- DAYBUE STIX, a new powder for oral solution, received US FDA approval in December 2025
- Broader commercial launch of DAYBUE STIX targeted for early Q2 2026
- DAYBUE STIX is expected to re-engage approximately 400 additional naïve and discontinued patients
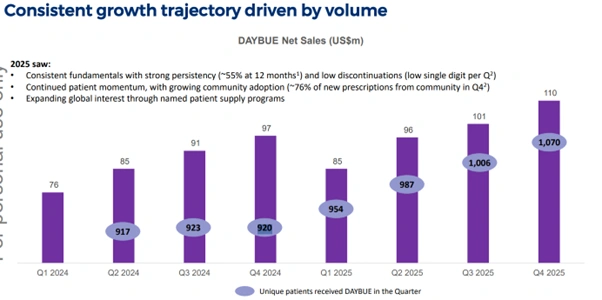
Figure 3: DAYBUE quarterly net sales growth and increasing patient volumes through 2024-2025 [Neuren Pharmaceuticals]
Royalty income to Neuren Pharmaceuticals ASX is estimated at $70 million to $77 million in CY2026, up 8–19% on CY2025.
Acadia Deal Economics Highlight Neuren Pharmaceuticals ASX Upside
The Acadia partnership underpins the Neuren financial highlights narrative. Payments received to date include:
- US$10 million upfront in 2018
- US$10 million following NDA acceptance in 2022
- US$40 million following the first US commercial sale in 2023
- US$50 million as one-third share of the Priority Review Voucher sold for US$150 million in 2024
- US$55 million in milestone payments related to Fragile X
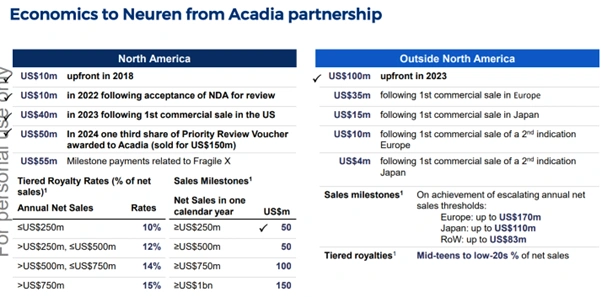
Figure 4: Royalty, milestone and tiered payment structure from the Acadia Pharmaceuticals partnership [Neuren Pharmaceuticals]
Additional sales milestones of up to US$350 million remain in North America alone, with up to US$363 million available across Europe, Japan and the rest of the world.
Global Market Access Opportunities in the Neuren Investor Presentation 2026
The Neuren financial highlights outlined trofinetide’s global regulatory progress:
- United States – approved March 2023, 6,000-9,000 estimated Rett patients
- Canada – approved October 2024, 600-900 estimated Rett patients; reimbursement not currently recommended
- Israel – approved January 2026
- Rest of World – named patient supply programs active in select countries via FarmaMondo
- Europe – 9,000–12,000 estimated Rett patients; active named patient programs via Clinigen; CHMP re-examination planned with potential outcome late Q2 2026
- Japan – 1,000–2,000 estimated Rett patients; Orphan Drug Designation granted; Phase 3 results expected Q4 2026 to Q1 2027; marketing application targeted for 2027
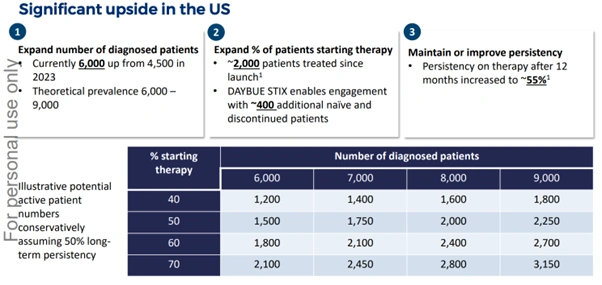
Figure 5: Phase 2 clinical trial results for NNZ-2591 in Pitt Hopkins syndrome showing meaningful improvements across key domains [Neuren Pharmaceuticals]
Each new market approval triggers additional milestone payments to Neuren and adds an ongoing royalty stream.
NNZ-2591 Pipeline Progress at Neuren Pharmaceuticals ASX
NNZ-2591 is Neuren’s wholly-owned next-generation compound. It targets the same IGF-1 upstream biological pathway as trofinetide and is being developed across six rare neurodevelopmental conditions and one brain injury indication. The Company holds worldwide rights to all NNZ-2591 indications outside of Rett and Fragile X, which are licensed to Acadia.
The two lead NNZ-2591 programs as of the Neuren investor presentation 2026:
Phelan-McDermid Syndrome (PMS)
- No FDA-approved treatments currently exist
- Phase 2 trial results were robustly positive: 16 out of 18 subjects improved on both co-primary endpoints, with statistical significance of P < 0.0001
- FDA alignment achieved on a single Phase 3 trial design and endpoints to support a New Drug Application
- Designations held: Orphan Drug (US and EU), Rare Pediatric Disease (US), Fast Track (US)
- Koala Phase 3 trial has commenced with the first 2 patients dosed and more than 70 patient referrals as of 27 February 2026
- Trial design: 160 patients randomised 1:1 over 13 weeks, double-blind, followed by 12-month open-label extension
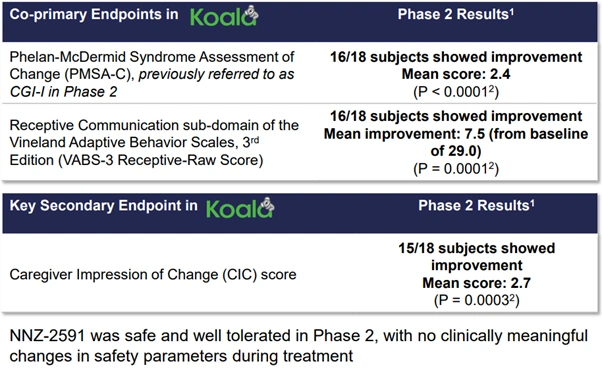
Figure 6: US market upside illustration highlighting patient diagnosis growth, therapy initiation and treatment persistence [Neuren Pharmaceuticals]
- No FDA-approved treatments currently exist
- Phase 2 trial: 9 out of 11 children showed improvement, with a mean CGI-I score of 2.6 (p = 0.0039)
- Improvements seen in communication, social interaction, cognition and motor abilities
- Designations held: Orphan Drug (US and EU), Fast Track (US), Rare Pediatric Disease (US)
- FDA open to flexible trial designs and patient-focused co-primary endpoints
- Next steps include assessing alternative trial designs and further FDA interaction
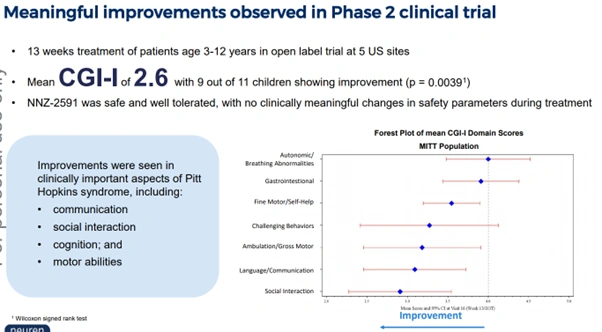
Figure 7: Co-primary and secondary endpoint outcomes from the Koala Phase 2 trial in Phelan-McDermid syndrome [Neuren Pharmaceuticals]
Share Price Performance
Neuren Pharmaceuticals Limited (ASX: NEU) last traded at $13.360 per share. The 52-week range is $8.610 to $22.985. Market capitalisation is approximately $1.70 billion.
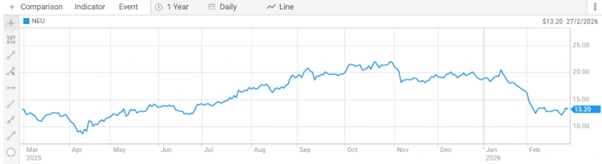
Figure 8: One-year share price performance of Neuren Pharmaceuticals Limited [ASX]
Investor Outlook
The Neuren investor presentation 2026 frames the Company as a commercially funded biotech with growing recurring income and multiple late-stage clinical catalysts ahead. Key watchpoints for investors:
- CY2026 DAYBUE net sales trajectory against Acadia’s US$460–490 million guidance
- DAYBUE STIX commercial launch in Q2 2026 and its impact on patient numbers
- European CHMP re-examination outcome expected late Q2 2026
- Japan Phase 3 trofinetide results expected Q4 2026 to Q1 2027
- Koala Phase 3 trial enrolment progress for PMS
- PTHS Phase 2/3 trial design finalisation and further FDA interaction
- HIE juvenile animal data generation and IND submission timeline
- AUD/USD exchange rate movement and its impact on reported royalty income in Australian dollars
Frequently Asked Questions
Q1. What does Neuren do and how does it make money?
Ans. Neuren develops drugs for childhood neurodevelopmental disorders. It earns revenue from royalties and milestones tied to DAYBUE (trofinetide), commercialised by Acadia, and is advancing its own drug NNZ-2591 across multiple rare conditions.
Q2. What were the key financial highlights in FY2025?
Ans. Royalty income rose 15% to $65m, NPAT was $30m (excluding one-offs), R&D spend was $36m, and cash reached $296m. Total DAYBUE income since launch is $510m.
Q3. How advanced is NNZ-2591?
Ans. The Phase 3 Koala trial for Phelan-McDermid syndrome has started, Pitt Hopkins Phase 2 data were positive, and FDA feedback supports further development, including for HIE.
Q4. Why is DAYBUE STIX important?
Ans. DAYBUE STIX is a new FDA-approved formulation that improves convenience and is expected to re-engage ~400 patients, supporting further royalty growth from 2026.