Altech Chemicals Ltd (ASX: ATC) presents CERENERGY® Batteries as the ideal grid storage alternative to Lithium-Ion Batteries (LIBs), overcoming the costs and challenges faced by LIBs.
Challenges with Lithium-ion Batteries
Fire explosion
Lithium-ion batteries come with a flammable liquid electrolyte and a flammable plastic separator. Overheating or overcharging the battery can lead to oxygen production inside the battery, which can lead to a thermal runaway or fire explosion, hard to put out.
Not Suitable for Cold/desert Climates
Lithium-ion batteries operate in a relatively narrow temperature range (between +15oC to +35oC). At lower temperatures, the liquid electrolyte in the battery turns more viscous, slowing lithium reactions. At 0°C, the battery capacity reduces to 70%.
Battery Lifespan
Lithium-ion degrades with each charge and discharge cycle. This is often due to detrimental side reactions and crystalline dendrite growth. The degradation can intensify when lithium-ion batteries operate outside the ideal temperature range. The grid storage lifespan of lithium-ion batteries is expected to be 7-10 years. Most electric vehicle (EV) manufacturers will guarantee eight years of battery life when the battery’s capacity drops below 70%.
Cost

The global lithium market is increasing, and the price of lithium has risen six-fold since the start of the year. This spike in price range puts upward pressure on the production costs for lithium-ion batteries.
Read More: Altech Chemicals Announces Launch Of CERENERGY® 60KWh Battery
Ethical Cobalt Supply
The global demand for cobalt is expected to surge to 220,000 mt by 2025. The Democratic Republic of Congo (DRC) is currently the biggest producer of cobalt, accounting for 70 percent of the global cobalt supply. However, child labor and human rights abuses in the DRC challenge the ethical supply chain concerns in the industry.
Graphite Supply
Graphite is expected to remain the dominant material until at least 2035. Hence there is a graphite deficit due to low supply, high demand, and cost.
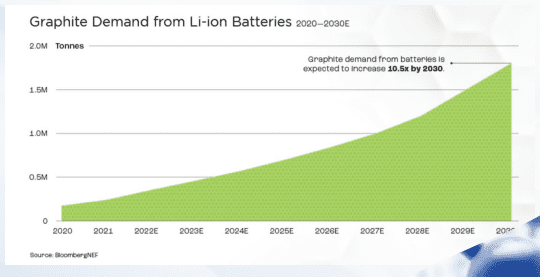
China produces 90% of the world’s graphite anode material, posing a geo-political supply chain risk to the industry.
Copper Crunch
Copper is a high-priced metal, and the EV industry requires 2.5 times more copper than an Internal Combustion Engine (ICE). More copper mines need to be built or expanded to meet the rising demand and scarcity.
With such challenges and concerns, what can be the ideal alternative to lithium-ion batteries?
Altech CERENERGY® Sodium Alumina Solid State Batteries
Altech’s CERENERGY® Sodium Alumina Solid State (SAS) Batteries can be the perfect alternative to lithium-ion batteries and their challenges.

WORKING OF CERENERY® SODIUM ALUMINA SOLID-STATE BATTERIES
Features of Altech’s CERENERY® SAS Batteries
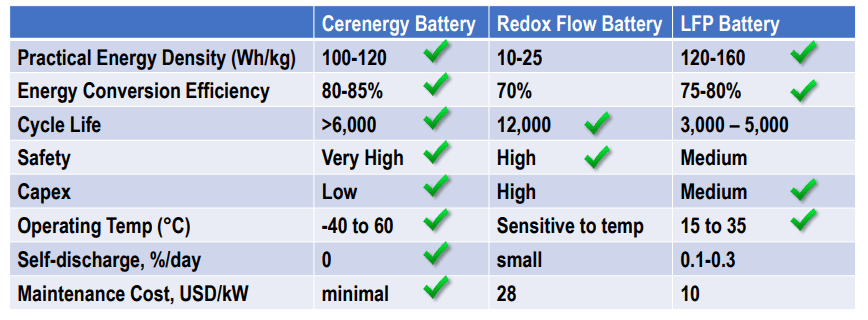
BATTERY TYPES COMPARISON
Fire-proof
CERENERGY® batteries have no volatile flammable electrolyte or plastic separator. The electrolyte is replaced with solid-state ceramic tubes. Due to this, there is no room for thermal runaway or oxygen production in the cathode, and it is safe to use in sensitive environments.
Large Operating Temperature Range
CERENERGY® batteries have an extensive temperature range between -40oC to +60oC and are ideal for operating in extreme cold and desert climates. Because the batteries have no liquid electrolyte, they can operate at ambient temperature without affecting the battery’s performance. These batteries are fully insulated, and the outer side of the battery module is at a “human touch” temperature.
Ultra-Long Battery Lifespan
Since CERENERGY® batteries have no liquid electrolyte, there is no deterioration or
loss of sodium ions or side reactions. The batteries have a lifespan of over 4,500 cycles and 15 years.
Lithium, Cobalt, Graphite, and Copper-free Batteries
- CERENERGY® batteries use sodium ions from common table salt. Common salt is cheap and always available, making it the perfect alternative to lithium.
- Instead of cobalt, the cathode consists of salt and nickel in a sodium aluminium chloride medium.
- CERENERGY® batteries have no anodes, so they are graphite and copper-free. They come with a self-forming anode at the (-) terminal of sodium when charged.